Key points:
- Hematopoietic stem cell transplantation (HSCT) can be a long-term treatment for multiple PIs, but has significant side effects and risks, including death, that people with PI, their loved ones, and their healthcare providers must discuss in detail before moving forward.
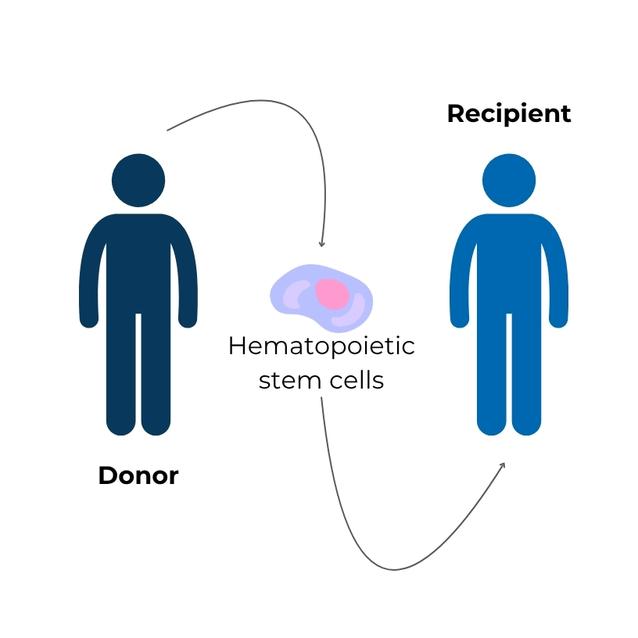
- Finding an appropriate donor is very important and can be the limiting step; however, modern technologies allow transplant doctors to find donors for almost all patients.
- Depending on the specific PI and the type of donor, the transplant process may include conditioning, which is a mix of chemotherapy and/or radiation designed to make room for the donor’s cells and prevent the recipient’s body from rejecting them.
- Choosing a transplant center is an important but personal decision and discussing options with an expert on the specific PI being treated may be helpful.
- HSCT cannot 'fix' problems outside of the blood or immune system and people who have had a successful HSCT can still pass PI-causing gene variants on to their children.
Find hematopoietic stem cell transplant clinical trials
See if you are eligible for clinical trials evaluating new approaches and protocols for treating PI with a hematopoietic stem cell transplant (HSCT).
Become a stem cell donor
Interested in becoming a stem cell donor? The National Marrow Donor Program (NMDP), formerly Be The Match, runs a U.S. stem cell registry that helps people who need HSCT find an appropriate donor. See if you qualify!
Preparing for allogeneic HSCT
Choosing a transplant center
You may or may not have options when it comes to choosing a transplant center. Sometimes health insurance providers require people to use certain specialists or certain facilities. Two resources for finding transplant specialists include the Primary Immune Deficiency Treatment Consortium (PIDTC) [1] and The National Marrow Donor Program [2]. Organizations that help advocate for specific conditions may also be a good resource.
Some people may be fortunate enough to have a transplant center near home, but many will have to travel to specialized centers for HSCT. This can be emotionally and financially challenging for families and caregivers. The time spent at a transplant center varies from center to center and from person to person. However, recipients and caregivers can generally expect to stay at or near the transplant center for at least 3-6 months before returning home. Allogeneic HSCTs are generally performed in a hospital, but once recipients are healthy enough, they are discharged. Even then, they have to return to the transplant clinic several times a week as they recover.
Individuals with PI and their families or caregivers usually meet members of the transplant team, which can include physicians, nurse practitioners, social workers, psychologists, nurses, and financial counselors. These team members help prepare recipients and their families for the impact the transplant will have on daily life, and they provide support and assistance with all aspects of the transplant.
Here is a list of questions to ask a transplant team before choosing them to perform the HSCT.
- How many cases of the specific PI has the hospital treated with HSCT? How are the patients doing/what are the outcomes?
- How many years has the hospital used HSCT to treat that PI?
- How many HSCT doctors are on staff?
- How much conditioning will be required before the HSCT?
- Which doctor or group of doctors will be in charge of the recipient’s care after discharge?
Individuals with PI and their families or caregivers should learn as much as they can about the HSCT procedure beforehand. The following questions can be used to both educate caregivers and evaluate the transplant center’s experience.
- What precautions should be taken prior to admission for transplant?
- How long will the recipient be in isolation?
- What isolation precautions are required before, during, and after transplant?
- What are the responsibilities of caregivers to keep isolation precautions in place?
- What are the isolation guidelines and expectations of members of the medical team and visitors when entering the recipient’s hospital room?
- Describe the complications related to HSCT for a person with this specific PI.
- If things go well during transplant, what is the average amount of time in the hospital and the average recovery time?
- What is expected of a caregiver while the recipient is in the hospital and after the recipient goes home?
- When can the recipient receive vaccines (live and nonviable) after the HSCT?
Staff at the hospital, working with the family or caregivers, will often work with insurance companies to get approval for the transplant.
Pre-transplant tests and procedures
HSCT recipients have a large number of tests and procedures done before transplant to make sure they are healthy enough to have a transplant. Healthcare providers may find hidden problems that need to be addressed prior to transplant, such as infections or organ function problems.
Immunologists and transplant providers typically order many blood tests, as well as imaging tests, such as computerized tomography (CT) scans, ultrasounds, and/or magnetic resonance imaging (MRI). Special tests of kidney function are also done. The heart is evaluated with an echocardiogram (ECG, EKG), which is an ultrasound of the heart. If recipients are old enough, pulmonary function testing will be done. Some may need procedures such as a bone marrow biopsy, lumbar puncture (also known as a spinal tap), bronchoalveolar lavage, or gastrointestinal endoscopy prior to allogeneic HSCT. All of these tests usually take a few weeks to complete.
Recipients and their families or caregivers may also meet with a fertility specialist, as chemotherapy used in conditioning can sometimes decrease the ability to have children in the future. These options will be different depending on the patient’s sex, age, and pubertal status, as well as the transplant center. For patients that have already undergone puberty, semen collection or egg harvesting is typically recommended. Of note, egg harvesting requires hormone stimulation and may take weeks and/or multiple attempts to be successful. It is possible a patient’s clinical status would not allow time for such a procedure to happen. For pre-pubertal patients, ovarian or testicular tissue cryopreservation, or long-term freezing, may be available. Though both of these are currently experimental, they offer the potential for future fertility.
Consent for transplant
Before having a transplant, a recipient and/or caregiver must give consent to proceed with the transplant. Members of the transplant team will meet with the recipient and/or caregiver to go over the process and the risks and benefits in detail, including the medications that will be used and their possible side effects. Recipients and caregivers should ask questions and be sure that they understand the proposed treatment before giving consent.
Hospital discharge
The recovery process for a recipient after HSCT takes at least several months, and will vary according to conditioning, the type of donor, any complications, and the individual hospital or transplant center’s criteria for discharge. Typically, once a recipient's donor cells are making enough neutrophils and platelets to keep the patient safe, they are ready for discharge. Recipients must not have fevers or bleeding, and they need to be able to take their medications as prescribed. Recipients or caregivers must be comfortable taking care of the central line, and they may also need to learn to give IV fluids, IV nutrition, and even some IV medications.
Once a recipient is discharged, they usually come back to the transplant clinic several times a week for lab work, examinations, and infusions. Often recipients are discharged to housing that is close to the transplant center, particularly if they are far from home. Recipients can still develop complications after discharge, including infections, organ damage from medications, and GVHD.
Once engraftment occurs, and the frequency of blood tests goes down, the central line may be taken out.
By Day +100, some recipients are ready to return to their own homes and can continue to follow up with their local healthcare providers. Recipients often still need to take several medications for the first year after transplant, including medications to prevent GVHD. Until they have a full recovery of immune cell numbers and are successfully weaned off of immunosuppressive medications, recipients should remain isolated at home. Visitors should remain restricted, and the recipient will not be allowed outside, or in public places, for several months. Neutrophils recover a few weeks after transplant, but T cells require many months and B cells may take 1-2 years.
Healthcare providers can do blood tests to see how an individual’s immune system is recovering and make recommendations about when it is safe to stop isolation. Once the immune system is fully recovered, recipients should receive routine re-vaccination following the transplant.
Latest HSCT resources
This page contains general medical and/or legal information that cannot be applied safely to any individual case. Medical and/or legal knowledge and practice can change rapidly. Therefore, this page should not be used as a substitute for professional medical and/or legal advice. Additionally, links to other resources and websites are shared for informational purposes only and should not be considered an endorsement by the Immune Deficiency Foundation.
Adapted from the IDF Patient & Family Handbook for Primary Immunodeficiency Diseases, Sixth Edition.
Copyright ©2019 by Immune Deficiency Foundation, USA
Sign up for updates from IDF
Receive news and helpful resources to your cell phone or inbox. You can change or cancel your subscription at any time.