Key points:
- Immunoglobulin (Ig) replacement therapy provides antibodies to protect people with primary immunodeficiency (PI) from infections.
- Ig replacement therapy needs to be tailored to each person’s medical condition and personal preferences, including dose, infusion frequency, and route of administration, to minimize infections while also avoiding side effects.
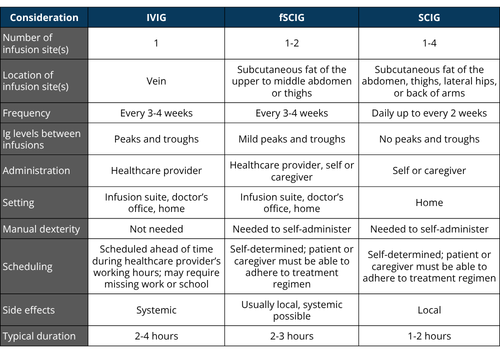
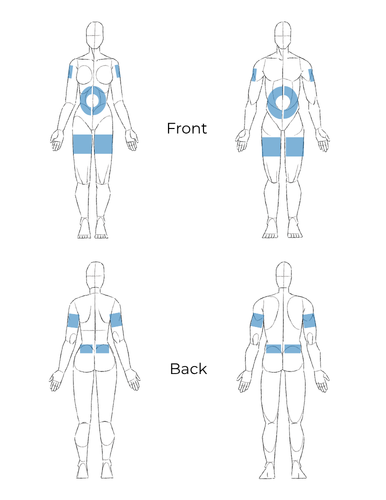
- Ig replacement therapy can be given intravenously (IVIG) or subcutaneously (SCIG or SubQ), and each route has its benefits and drawbacks.
- Most people with PI need Ig replacement therapy for life.
Find Ig replacement therapy clinical trials
See if you qualify to participate in clinical trials evaluating Ig replacement therapy.
Ig manufacturing and safety
All Ig products are made from donated human plasma. There are multiple steps in the production process to make sure the end product is safe.
First, plasma donation centers screen all plasma donors by asking for a detailed history of infections and risk behaviors, such as intravenous drug use. Donors also complete testing for certain viruses using very sensitive tests. Donors can only give plasma if they pass this screening. If the screening identifies any risk factors, the person is excluded from donation, or deferred. The FDA requires centers to maintain a central list of people who have been deferred to prevent them from donating at other centers or in the future [4].
As an added protection, plasma donors must return to donate a second time within a few weeks of their first donation. They are then rescreened for viruses and risk factors. If a donor does not return within that timeframe or the second screening identifies risk factors, the center throws away the plasma from their first donation.
Plasma centers then test each individual plasma donation for germs before pooling it with plasma from thousands of other donors. Once the plasma is pooled, the entire pool is tested for HIV and hepatitis A, B, and C viruses. Different separation and filtration methods help pull out IgG antibodies from the pooled plasma, and manufacturers test for viruses at multiple points throughout this process.
In the mid-1990s, some IVIG users developed non-A, non-B hepatitis (now known to be caused by the hepatitis C virus (HCV)). These rare infections caused manufacturers to add an extra step in the manufacturing process to dissolve, or inactivate, these types of viruses. More recently, manufacturers have added a final ultrafiltration or depth filtration step to remove prions, which are infectious proteins that cause diseases like mad cow disease. Notably, HIV, which is destroyed in the first step in the production of Ig, has never been spread through the use of any Ig product.
Usually, one lot of Ig product is made from the plasma of about 10,000 donors. This pooling makes sure that all Ig lots contain a broad variety of antibodies that are found in the general population to specific germs and routine vaccinations, which then provide protection to people with PI. By law, all Ig products licensed in the U.S. must be made from source plasma that has been collected in the U.S. [5].
Starting Ig replacement therapy
The goal of Ig replacement therapy for PI—no matter the setting, dose, or route of administration—is to provide protection from germs. Being consistent with Ig replacement therapy is very important for reaching this goal. Any challenges, real or potential, need to be addressed to make sure that the person with PI remains on schedule for their infusions.
Healthcare providers set someone’s initial Ig dose based on their weight. Typically, a starting dose is between 400-600 mg/kg/month. Healthcare providers then adjust the dose to minimize the frequency and severity of infections while also minimizing any side effects from the medication.
It is important to track any side effects during or after infusions and tell the prescribing healthcare provider about them. People should also report any side effects to the product manufacturer and MedWatch at the FDA in case there is a problem with the product lot [6].
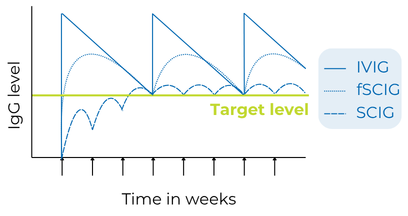
Monitoring IgG levels over time is important to track the person’s response to therapy. The goal is to keep the Ig in the bloodstream above a target level, which differs for each person, even at its lowest point right before the next infusion, which is known as the trough level.
It is also important to remember several things when starting Ig replacement therapy:
- Ig replacement therapy can’t prevent all infections.
- After starting Ig replacement therapy, you may still get infections, especially if you are not yet at the dose that works best for you. However, the frequency and severity of infections should significantly decrease so that permanent organ damage, like bronchiectasis, does not develop.
- Because the antibody levels in Ig products mirror what plasma donors have been vaccinated against and exposed to, Ig products may not provide strong or reliable protection against fast-changing germs, such as the newest COVID-19 or influenza virus strains. Antibody levels to whooping cough (pertussis) also vary among products and may not be protective.
- If you are still getting multiple or severe infections after being on a steady Ig replacement therapy regimen, talk to your prescribing healthcare provider about increasing your Ig dose or the frequency of your infusions.
- One size does not fit all. A customized Ig replacement therapy regimen must be developed for each person and modified as needed to achieve treatment goals and meet the person’s needs.
- Once someone has been diagnosed with an antibody deficiency, they will likely need to be on Ig replacement therapy for life. Exceptions are people who are treated with Ig replacement therapy temporarily in order to prevent infections while waiting for HSCT or gene therapy. In addition, some children may no longer need Ig replacement therapy if they “outgrow” their antibody deficiency as their immune system matures.
- Ig can inactivate live vaccines. If someone is on Ig replacement therapy at the time they receive a vaccine, the antibodies in Ig products can make the vaccine ineffective, especially live vaccines. The exceptions are seasonal vaccines, like the flu and COVID-19 vaccines, or vaccines for germs not typically seen in the U.S., like typhoid.
- Ig can interfere with antibody-based laboratory tests. If someone is on Ig replacement therapy, antibody-based (serology) tests, which are often used to check for exposure to a particular germ like the hepatitis B virus, will not be accurate. They will measure the antibodies in the Ig product rather than the antibodies the person makes. Healthcare providers should use antigen or molecular tests instead.
- In some cases, healthcare providers may need to reevaluate a diagnosis. This must be done cautiously, because Ig replacement therapy has to be paused for at least four months to get accurate results for antibody-based laboratory tests.
Tests to complete beforehand
Before starting Ig replacement therapy, it is important that a healthcare provider completes all laboratory tests needed to show that the person’s Ig levels are low and/or that they do not make working antibodies. These tests typically include a serum immunoglobulin test and a vaccine challenge test.
Insurance companies often require these test results as part of their prior authorization paperwork and will not approve Ig replacement therapy without them. You should keep a copy of the results with your other important medical information in case your insurance changes or you become eligible for Medicare. Medicare will not cover Ig replacement therapy without serum immunoglobulin and vaccine challenge test results. Once someone starts Ig replacement therapy, it is not possible to get accurate results for these important tests without stopping treatment for several months. Stopping treatment to redo these tests places you at risk of getting infections.
Insurance considerations
Ig replacement therapy typically requires the prescribing healthcare provider to get prior authorization from the person’s health insurance company. In addition, health insurance providers may not cover all Ig products and sometimes place other restrictions on Ig replacement therapy, such as limiting where a person can get their infusions. Be sure to check with your insurance plan to determine which routes of administration and/or Ig products are covered and whether there are any other restrictions on Ig replacement therapy.
For people on Medicare, how Ig replacement therapy is covered depends on the ICD-10 diagnostic code your provider uses to prescribe your medication. The Centers for Medicare and Medicaid Services (CMS) only allows certain ICD-10 diagnostic codes to be used for Ig replacement therapy covered under Medicare Part B; see below. Other diagnostic codes may be covered under Medicare Part D, which is the optional prescription coverage. Before purchasing a Medicare Part D plan, be sure it covers Ig replacement therapy if your healthcare provider does not use an ICD-10 covered under Part B.
Products approved for IVIG and/or SCIG in the U.S.
All products can be stored either refrigerated or at room temperature (not to exceed 77°F or 25°C) for at least four weeks; check package inserts to see if your specific product can be stored for longer. Once a product has reached room temperature, it should not be returned to refrigerated temperature.
Products should never be frozen and should be discarded if they become frozen. Refer to product package inserts for additional storage details.
Alyglo
Administration: IVIG
Concentration: 10% (100 mg/mL)
Approved for: Ages 17+
Report side effects/adverse reactions at medicalinfo@gcbiopharmausa.com or 1-833-426-6426.
Asceniv
Administration: IVIG
Concentration: 10% (100 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at PV@admabio.com or 1-800-458-4244, option 2.
Bivigam
Administration: IVIG
Concentration: 10% (100 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at PV@admabio.com or 1-800-458-4244, option 2.
Cutaquig
Administration: SCIG
Concentration: 16.5% (165 mg/mL)
Approved for: Ages 2+
Octapharma and Pfizer both distribute Cutaquig. Each company has its own product website and may have its own copay assistance program. If your infusion center or hospital supplies Cutaquig, it is typically distributed by Octapharma. If you get Cutaquig through a specialty pharmacy/for home care, it is distributed by Pfizer.
Report side effects/adverse reactions at 201-604-1137.
Cuvitru
Administration: SCIG
Concentration: 20% (200 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at pvsafetyamericas@takeda.com or 1-877-825-3327.
Flebogamma DIF
Administration: IVIG
Concentration: 5% (50 mg/mL) or 10% (100 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at https://www.grifols.com/en/notification-of-adverse-reaction.
Gammagard Liquid
Administration: IVIG or SCIG
Concentration: 10% (100 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at pvsafetyamericas@takeda.com or 1-877-825-3327.
Gammagard Liquid ERC
Administration: IVIG or SCIG
Concentration: 10% (100 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at pvsafetyamericas@takeda.com or 1-877-825-3327.
Gammagard S/D
NOTE: Takeda is discontinuing Gammagard S/D in December 2027.
Administration: IVIG
Concentration: 5% (50 mg/mL) or 10% (100 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at pvsafetyamericas@takeda.com or 1-877-825-3327.
Gammaked
Administration: IVIG or SCIG
Concentration: 10% (100 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at 1-855-353-7466 or https://www.kedrion.us/ pharmacovigilance/.
Gammaplex
Administration: IVIG
Concentration: 5% (50 mg/mL) or 10% (100 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at 1-855-353-7466 or https://www.kedrion.us/ pharmacovigilance/.
Gamunex - C
Administration: IVIG or SCIG
Concentration: 10% (100 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at https://www.grifols.com/en/notification-of-adverse-reaction.
Hizentra
Administration: SCIG
Concentration: 20% (200 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at Adverse.Events.Global@cslbehring.com or 1-866-915-6958.
HyQvia
Administration: Facilitated SCIG
Concentration: 10% (100 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at pvsafetyamericas@takeda.com or 1-877-825-3327.
Octagam
Administration: IVIG
Concentration: 5% (50 mg/mL) or 10% (100 mg/mL)*
* Octagam 10% is approved for chronic immune thrombocytopenia and dermatomyositis.
Approved for: Ages 6+
Octapharma and Pfizer both distribute Octagam. Each company has its own product website and may have its own copay assistance program. If your infusion center or hospital supplies Octagam, it is typically distributed by Octapharma. If you get Octagam through a specialty pharmacy/for home care, it is distributed by Pfizer.
Report side effects/adverse reactions at 201-604-1137.
Panzyga
Administration: IVIG
Concentration: 10% (100 mg/mL)
Approved for: Ages 2+
Octapharma and Pfizer both distribute Panzyga. Each company has its own product website and may have its own copay assistance program. If your infusion center or hospital supplies Panzyga, it is typically distributed by Octapharma. If you get Panzyga through a specialty pharmacy/for home care, it is distributed by Pfizer.
Report side effects/adverse reactions at 201-604-1137.
Privigen
Administration: IVIG
Concentration: 10% (100 mg/mL)
Approved for: all ages
Report side effects/adverse reactions at Adverse.Events.Global@cslbehring.com or 1-866-915-6958.
Qivigy
Administration: IVIG
Concentration: 10% (100 mg/mL)
Approved for: Ages 18+
Report side effects/adverse reactions at 1-855-353-7466 or https://www.kedrion.us/ pharmacovigilance/.
Xembify
Administration: SCIG
Concentration: 20% (200 mg/mL)
Approved for: Ages 2+
Report side effects/adverse reactions at https://www.grifols.com/en/notification-of-adverse-reaction.
Yimmugo
Administration: IVIG
Concentration: 10% (100 mg/mL)
Approved for: Ages 2+
Kedrion distributes Yimmugo in the U.S.
Report side effects/adverse reactions at 1-855-353-7466 or https://www.kedrion.us/ pharmacovigilance/.
This page contains general medical and/or legal information that cannot be applied safely to any individual case. Medical and/or legal knowledge and practice can change rapidly. Therefore, this page should not be used as a substitute for professional medical and/or legal advice. Additionally, links to other resources and websites are shared for informational purposes only and should not be considered an endorsement by the Immune Deficiency Foundation.
Adapted from the IDF Patient & Family Handbook for Primary Immunodeficiency Diseases, Sixth Edition.
Copyright ©2019 by Immune Deficiency Foundation, USA
Sign up for updates from IDF
Receive news and helpful resources to your cell phone or inbox. You can change or cancel your subscription at any time.